| Issue |
EPJ Nuclear Sci. Technol.
Volume 3, 2017
|
|
|---|---|---|
| Article Number | 30 | |
| Number of page(s) | 7 | |
| DOI | https://doi.org/10.1051/epjn/2017027 | |
| Published online | 03 November 2017 | |
https://doi.org/10.1051/epjn/2017027
Regular Article
Growth of micrometric oxide layers to explore laser decontamination of metallic surfaces
Den − Service d'Etudes Analytiques et de Réactivité des Surfaces (SEARS), CEA, Université Paris-Saclay,
91191
Gif sur Yvette, France
* e-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
10
July
2017
Received in final form:
9
October
2017
Accepted:
10
October
2017
Published online: 3 November 2017
Abstract
The nuclear industry produces a wide range of radioactive waste in terms of hazard level, contaminants and material. For metallic equipment like steam generators, the radioactivity is mainly located in the oxide surface. In order to study and develop safe techniques for dismantling and for decontamination, it is important to have access to oxide layers with a representative distribution of non-radioactive contaminants. In this paper we propose a method for the creation of oxide layers on stainless steel 304L with europium (Eu) as contaminant. This technique consists in spraying an Eu-solution on stainless steel samples. The specimens are firstly treated with a pulsed nanosecond laser after which the steel samples are placed in a 873 K furnace for various durations in order to grow an oxide layer. The oxide structure and in-depth distribution of Eu in the oxide layer were analyzed by scanning electron microscopy coupled to an energy-dispersive X-ray microanalyzer, as well as by glow discharge optical emission or mass spectrometry. The oxide layers were grown to thicknesses in the range of 200 nm–4.5 μm depending on the laser treatment parameters and the heating duration. These contaminated oxides had a ‘duplex structure’ with a mean concentration of the order of 6 × 1016 atoms/cm2 (15 μg/cm2) of europium in the volume of the oxide layer. It appears that europium implementation prevented the oxide growth in the furnace. Nevertheless, the presence of the contamination had no impact on the thickness of the oxide layers obtained by preliminary laser treatment. These oxide layers were used to study the decontamination of metallic surfaces such as stainless steel 304L using a nanosecond pulsed laser.
© L. Carvalho et al., published by EDP Sciences, 2017
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
In the nuclear industry, dismantling is a major issue. The contaminated surfaces vary widely in terms of characteristics such as shape (pipes, plane surfaces, etc.), conditions of oxide growth (temperature, atmosphere, etc.) and nature of contaminants. In regard to metallic components such as pipe systems, radio nuclides are typically found in the oxide layer, which grows during the operations of the nuclear facility. The cleaning of contaminated surfaces is currently based on chemical and mechanical processes. However, a few problems remain unsolved. These techniques usually generate secondary wastes, contaminated effluents requiring long-term storage and dismantling by workers who thus become exposed to occasional radiation.
In this context, studies have been carried out on the cleaning of contaminated surfaces such as painted concrete [1,2] and stainless steel [3,4] by excimer or fiber lasers. In particular, the CEA (French Alternative Energies and Atomic Energy Commission) has developed a prototype for the cleaning of painted surfaces called Aspilaser [2]. The principle of this technique is laser ablation of the surface in order to remove the oxide layer and collect the radioactive particles with a High Efficiency Particulate Air (HEPA) filter for storage. In nuclear facilities, the use of a laser for the cleaning of contaminated surfaces has many advantages such as the possibility of remote treatment without worker exposition. As a dry process, the secondary wastes generated by laser cleaning are the ablated particles and the HEPA filter, that can be stored in a small volume without risk of dispersion.
A previous study [3,5] has highlighted the need to create oxide layers in a controlled manner for the study of the efficiency of laser decontamination and to optimize the cleaning process. The aim of growing oxide layers on metallic surfaces with a non-radioactive contaminant is to study and develop decommissioning techniques without the problems of availability of radioactive samples and the constraints of working in hot laboratories.
The goal of our work has been to find a way of creation of oxide layers with desired characteristics (thickness, Eu-concentration) in order to explore laser cleaning. This paper presents the results of growing a micrometric oxide layer contaminated with a non-radioactive element and the preliminary test of laser cleaning.
2 Oxidation of stainless steel 304L with a non-radioactive marker
We have investigated a method to form a reproducible micrometric oxide layer on stainless steel AISI 304L (304L SS) with a volume-related distribution of europium (Eu) in the layer thickness. This distribution was chosen in order to consider Eu as a simulation of a radioactive element (actinide). Thus, the efficiency of decontamination can be determined by measuring the Eu concentration before and after laser decontamination. The initial composition of the 304L SS samples was analyzed via glow discharge mass spectrometry (GDMS) and no trace of Eu was found with a detection limit of 0.1 mg/kg (0.1 ppm) of metal.
2.1 Procedure
Specimens (30 mm × 30 mm × 3 mm) were cut by a water jet from a cold-rolled 304L stainless steel sheet. The coupons were cleaned in an ultrasound bath with ethanol. The average surface roughness has been measured by optical profiler and estimated to be Ra = 0.46 ± 0.13 μm. The contamination was generated before oxidation by deposition of a solution of Eu(NO3)3 in water at an Eu-concentration of 4000 mg/L. The solution was sprayed with a nebulizer and the flow was controlled by a pumping device. The samples were heated using a hot plate at 473 K until the deposition had dried. It was estimate that the deposited amount of Eu was 0.5 up to 1.0 mg/cm2 (1.6–3.2 × 1019 atoms/cm2). Certain samples without contamination served as control samples.
Pre-oxidation was carried out by laser treatment in air at atmospheric pressure. The setup comprised an ytterbium fiber laser (Gaussian beam, M2 = 2.1, λ = 1064 nm, mean power up to 20 W, repetition rate v = 20 kHz, pulse duration τ = 110 ns at FWHM), a scanning system, a focusing lens (f = 420 mm) resulting in a laser beam waist radius ω = 84 ± 2 μm (at 1/e2 intensity level) and an air aspiration system. For the oxidation regime, the samples were mounted on a micro control assembly and placed at the focal lens. The power of the beam varied between 5 and 18 W in order to study a maximal fluence per pulse (F) in the range 2.2–7.8 J/cm2 (maximal fluence on the beam axis).
The laser treatment was conducted by horizontal and vertical scanning (Fig. 1). The horizontal scanning was characterized by the scanning speed V in order to define the step Δx between two consequent laser spots. The vertical scanning was defined by the vertical step Δy between two parallel lines.
The scan speed and the distance between lines were chosen to provide a high spatial overlapping between two successive spots from 80–95%. The overlapping percentage was calculated using the laser spot beam diameter at 1/e2. The laser treatment was defined by the maximum beam fluence F per pulse and the delivered energy amount E per surface unit of the sample after the treatment. F is calculated as follows:
 (1)
where Epulse is the energy per pulse in joule and S is the surface of the laser spot in cm2. The energy per pulse is the laser average power (P) divided by the repetition rate v of 20 kHz. The average energy per surface unit delivered by the laser source on the sample E is determined as follows:
(1)
where Epulse is the energy per pulse in joule and S is the surface of the laser spot in cm2. The energy per pulse is the laser average power (P) divided by the repetition rate v of 20 kHz. The average energy per surface unit delivered by the laser source on the sample E is determined as follows:
 (2)where Δt is the treatment duration and Ss is the total treated surface in cm2. A part of the energy delivered by the laser source is lost by reflection on the surface. The absorption coefficient of the AISI 304L samples at λ = 1064 nm was determined by integrating sphere and is estimated to be 29 ± 2%. In this work, we study oxide layers formed by a delivered energy E in a range from 50 to 600 J/cm2.
(2)where Δt is the treatment duration and Ss is the total treated surface in cm2. A part of the energy delivered by the laser source is lost by reflection on the surface. The absorption coefficient of the AISI 304L samples at λ = 1064 nm was determined by integrating sphere and is estimated to be 29 ± 2%. In this work, we study oxide layers formed by a delivered energy E in a range from 50 to 600 J/cm2.
After laser treatment, the oxidized samples were placed in a furnace at 873 K for 50 or 100 hours in air at atmospheric pressure and their cooling was done in the furnace after it was switched off. Such a furnace treatment makes it possible to grow the oxide layer to a thickness up to 4.5 μm depending on the oxidation time in the furnace.
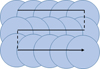 |
Fig. 1 Pattern of the scanning regime. The circles represent the consecutive laser spots on the treated surface and the continuous arrow shows the laser beam path. |
2.2 Characterization of the oxide coatings
The oxidation procedure resulted in the creation of Eu-contaminated oxide layers with a thickness from 200 nm to 4.5 μm depending on the set of laser parameters and the duration of the heating. The obtained oxide layers were characterized and analyzed by glow discharge optical emission spectrometry (GD-OES), GDMS, scanning electron microscopy (SEM) coupled with an energy dispersive X-ray analyzer (EDX), X-ray diffraction (XRD) and micro-Raman spectroscopy.
The Eu-contaminated layer formed after the pre oxidation by laser was analyzed by GD-OES and the in-depth distribution of the elements is presented in Figure 2. One can see that the laser treatment resulted in the creation of an oxide layer (mostly Fe- and Cr-oxide) of a thickness of 500 nm with an Eu distribution in the whole oxide thickness and with a mean Eu-concentration up to 3.8 wt.% or 6 × 1016 atoms/cm2.
A GD-OES profile and a SEM image (back scattering electrons) of a representative oxide layer (E = 600 J/cm2, oxidation time in the furnace of 50 h) are shown in Figures 3 and 4.
The oxidation in the furnace led to a layer of 4.1 ± 1.4 μm with a contamination of 0.40 ± 0.13 wt.% of Eu in the whole thickness. The thermal treatment only induced diffusion of contaminant, thus the concentration remains at 6 × 1010 atoms/cm2. In both Figures 2 and 3, the distribution of Eu followed the oxygen distribution, signifying that the contamination was strictly retained in the oxide layer, and not detected in the bulk of the metal. The low detection limit of GD-OES (100 μg/g for Eu) allow us to highlighted the presence of Eu in the whole oxide.
Complementary investigation has been achieved by SEM coupled with EDX (Fig. 4). This technique allowed us to have access to the oxide thickness and to its chemical composition. However, in the conditions of observation (acceleration tension of 15 keV and a current of 2 nA), the EDX analysis is made on a volume of 1 μm3 and with a detection limit for Eu of 1% in weight, above the mean concentration of Eu in the oxide. For those reasons, mostly ferrous oxide was detected and the white parts in the SEM image of the cross section (Fig. 4) only matched the Eu-rich zones (10% of weight). The resulting contamination level for Eu appeared to be also lower than the detection limit of EDX, XRD and micro-Raman methods. The monitoring of Eu was decided to be conducted by GD-OES and GDMS, which have detection limits of 100 μg/g and 0.1 μg/g for Eu, respectively.
For thick oxide layers, the GD-OES profiles (Fig. 3) and SEM image (Fig. 5) led to the assumption of the presence of a thicker outer layer mainly composed of Fe-rich oxide and an inner one enriched in Cr. The micro-Raman spectroscopy (laser wavelength λ = 532 nm) and XRD analysis (grazing incidence, θ = 1°) of this sample showed mainly the creation of hematite Fe2O3 and a mixed oxide of Fe and Cr (Fe3O4/FeCr2O4). This structure appeared to be similar to the oxide layers in industrial nuclear conditions [6]. The oxide phases identified by XRD in samples with and without Eu were the same. Moreover, XRD analysis was not able to identify any specific phases with Eu like Eu-rich oxide because the concentration in Eu was below the detection limit of this technique.
Our result differed from the one in [7], where the authors proceeded with furnace oxidation of 304L SS specimens contaminated with ytterbium. In the study of Riffard et al., the contaminated layer was thinner and the oxide was composed mainly of a Cr-oxide: Cr2O3. However, our experiments showed the influence of rare earth metals as inhibitors of oxidation in the furnace, which was in accordance with other studies [7–9]. Thanks to our procedure, this blocking behavior of Eu is avoided by a previous laser scanning. The formation of a first oxide layer with Eu allows the oxide growth in the furnace.
Our investigations with the objective to find the oxidation condition to control the thickness of the oxide layer led us to test different laser regimes and furnace oxidation durations.
 |
Fig. 2 GD-OES profile of an Eu-contaminated oxide layer on 304L SS after a preliminary laser treatment (9 W, overlapping of 95%, E = 600 J/cm2) and without furnace treatment. The oxide thickness was estimated to be 500 nm. |
 |
Fig. 3 GD-OES profile of an Eu-contaminated oxide layer on 304L SS after preliminary laser treatment (9 W, overlapping of 95%, E = 600 J/cm2) and oxidation in the furnace (50 h, 873 K). The oxide thickness was estimated to be 4.5 μm. The scale of Eu-distribution was multiplied by 10 in order to be readable. |
 |
Fig. 4 SEM image (5 000x) of a cross section of a 4.5 μm thick Eu-contaminated oxide layer on 304L SS after a preliminary laser treatment (9 W, overlapping of 95%, E = 600 J/cm2) and oxidation in the furnace (50 h, 873 K). |
 |
Fig. 5 EDX map of a cross section of a 4.5 μm thick Eu-contaminated oxide on 304L SS after a preliminary laser treatment (9 W, overlapping of 95%, E = 600 J/cm2) and oxidation in the furnace (50 h, 873 K). |
2.3 Effect of oxidation parameters
The first step of our study was to determine the influence of the laser parameters on the preliminary oxide layer. To do so, we varied the delivered energy amount of the laser on the sample surface from 50 to 300 J/cm2 by using several laser powers (5, 9, 14 and 18 W) and 80 or 90% of spatial overlap. It was noticed that the preliminary oxide thicknesses ranged from 20 to 140 nm (Fig. 6). The oxide layers seemed to grow in a linear manner with the delivered energy amount.
Moreover, in the case of the treatments at 18 W/80% overlap and 5 W/90% overlap (40 nm), the oxides were quite similar in thickness although the fluence per pulse differed. This result highlights the fact that the principal oxidation parameter was the delivered energy amount E and not the fluence per pulse or the spatial overlap. The same experiments were performed without Eu-contamination and it appeared that the presence of Eu did not interfere with the oxidation by laser.
In order to span a large variety of laser treatments, the laser fluence was varied from 2.2 to 7.8 J/cm2 per pulse. In the case of a nanosecond pulse, the laser beam energy is absorbed by the surface due to electron excitation and transmitted to the metal lattice by collisions. This implies a localized heating of the metal surface, its oxidation and, possibly, its melting. The creation of the oxide during the laser scanning modifies the physical properties and the interaction regime between the laser and the surface. Interferences between incoming and outgoing beams can occur because of the presence of this semi-transparent oxide and implies temperature oscillations [10]. According to [11], the theoretical laser melt threshold fluence can be estimated by:
 (3)
where κ = 13 W/m·K is the thermal conductivity, θ = 1678 K is the melting temperature, τ = 110 ns is the laser pulse duration and D = 3.8 × 10‑6 m2/s, the thermal diffusivity of 304L stainless steel [12]. This fluence represents the minimum amount absorbed by the surface that leads to its melting. In our study, Φm = 0.36 J/cm2. The absorptivity of the surface was A = 0.35 and the melt threshold fluence could thus be estimated to 1 J/cm2 which was lower than the laser fluences used for the laser oxidation. SEM observations (Fig. 7) of single impacts of a laser pulse under the pre oxidation conditions showed proof of fusion of the surface during the treatment.
(3)
where κ = 13 W/m·K is the thermal conductivity, θ = 1678 K is the melting temperature, τ = 110 ns is the laser pulse duration and D = 3.8 × 10‑6 m2/s, the thermal diffusivity of 304L stainless steel [12]. This fluence represents the minimum amount absorbed by the surface that leads to its melting. In our study, Φm = 0.36 J/cm2. The absorptivity of the surface was A = 0.35 and the melt threshold fluence could thus be estimated to 1 J/cm2 which was lower than the laser fluences used for the laser oxidation. SEM observations (Fig. 7) of single impacts of a laser pulse under the pre oxidation conditions showed proof of fusion of the surface during the treatment.
A laboratory-developed analytical model [13] was used to simulate the laser heating of a metallic surface by nanosecond pulse radiation. The temperature spatial distribution or evolution in time could be determined. The simulation of a single impact of a 1064 nm-pulse (with a duration of 110 ns at a frequency of 20 kHz on a 304L stainless steel substrate) was performed for different laser fluences. At low fluence (1 J/cm2), it was seen that the temperature at the surface rose to its maximum of 1650 K after 150 ns and after 500 ns, the metal temperature was 750 K (Fig. 8). We can assume that the increase of the laser pulse energy can lead to the melting temperature (θ = 1678 K). The simulation condition (F = 1 J/cm2) is coherent with the melt threshold calculated in equation (3). When the maximum temperature is reached, the temperature profile in depth shows that the thermally affected zone is sub micrometric, like the resulting oxide layers.
The surface temperature calculated 50 μs (pulse repetition time) after a laser shot of F = 1 J/cm2 is still at 330 K. Thus, the pulse energy accumulation at the surface metal has a weak influence on the final surface temperature after one single pulse. Nevertheless, the simulation of multi impacts is needed to determine this influence during the laser treatment. Moreover, during laser scanning, the step between two pulses is less than 10% of the beam dimension. Thus we can consider that the treatment is performed on a surface which was already disturbed by the previous pulse. We will have to take into account the oxidation by the multi pulses on a substrate of 304L stainless steel with an oxide layer. For this purpose, a model of laser heating of an oxide layer on metallic surface has been developed in our laboratory and will be used [13]. More simulations need to be carried out but we can suppose that the oxidation is made from a liquid layer. This state allowed Eu to penetrate into the metal bulk and led to a fixed contamination of the sample surfaces.
The influence of the treatment duration on the oxide layer characteristics was studied. Contaminated samples (2.8 × 1019 atoms/cm2 of Eu) were first preliminary treated by laser (14 W, 90% overlap) and oxidized in a furnace at 873 K for duration from 25 to 200 h. The same experiments were performed with 304L samples without contamination. The preliminary oxide layer obtained by laser was 100–150 nm thick. The Figure 9 shows two main phenomena. On one hand, the contamination of Eu prevents the oxidation as expected in the literature [7–9]. On the other hand for blank samples, the oxide grows following a parabolic kinetics [7]. For the parabolic part of the curve, the parabolic rate is k = 6.14 ± 0.75 × 105μm2/s. Unlike the laser treatment, the oxidation by furnace is influenced by Eu-contamination. Indeed the experiments presented Table 1 shows that the increase Eu- concentration implies the limitation of the thickness of the oxide.
Even with the behavior of oxidation inhibitor of Eu, our oxidation procedure allowed us to continue the oxidation process. Given the influence of the main parameters (delivered laser energy, furnace oxidation duration or Eu concentration), the thickness of the resulting oxide layer could be controlled and adjusted depending on the study.
 |
Fig. 6 Oxide thicknesses resulting from different laser irradiations at 5, 9, 14 and 18 W and 80% overlap (in blue) or 90% overlap (in orange). |
 |
Fig. 7 SEM image (x 700) of a single impact on 304L SS after a different laser treatment: (a) 9 W (4.1 J/cm2), (b) 14 W (6.3 J/cm2). The red circles represent the thermally affected zone of the sample surface. |
 |
Fig. 8 Temperature evolution at the surface on the laser beam axis during a single laser pulse of 110 ns for a fluence of 1 J/cm2 on a blank AISI 304L substrate. |
 |
Fig. 9 Oxide thicknesses for different oxidation durations of Eu-contaminated samples (blue) and blank samples (orange). |
Oxide thickness after laser treatment (237 J/cm2 total delivered) and a furnace oxidation (100 h, 873 K) with different contamination concentration in Eu.
3 Laser decontamination of oxidized metal surfaces
According to the literature, the decontamination of metallic surfaces by excimers [4] and ytterbium fiber laser [3,5,14] was successful and demonstrated a decontamination percentage of up to 99% for thick oxide layer on stainless steel. The oxide layer was removed efficiently for the first passes but in a second phase, the decontamination stagnated. It was assumed that this limitation of the laser cleaning was the result of two phenomena: on the one hand, the trapping of the oxide and contaminants into micro-cracks of the materials preventing the laser beam from accessing the complete decontamination, and on the other hand, the heating of the surface during the laser treatment leading to the penetration of the contaminants in the metal bulk.
For decontamination of the oxide layer¸ the laser setup was the same as the pre-oxidation one. But the fluence applied to ablate the oxide layer was in the range of 4–8 J/cm2 with a spot overlapping of 80%. As shown in Figure 10, the visual appearance of the sample before and after decontamination is different. After cleaning treatment, the metallic aspect of the sample is restored. The samples were analyzed by GD-OES in order to determine the thickness of the oxide layer, but since the remaining Eu-concentration after cleaning was close to the detection limit of GD-OES, the GD-MS method was used (Fig. 11).
Figure 11 presents the GD-MS profiles for an oxide layer obtained after preliminary laser treatment (9 W, overlapping of 95%, E = 608 J/cm2) and furnace oxidation (50 h, 873 K) without and with decontamination treatment. The oxide layer was 3 μm-thick and had an Eu-contamination of 0.1 wt.% (Fig. 11a). It was seen that after 1 pass at 18 W and 80% overlapping, the resulting Eu-concentration was less than 0.02 wt.% (Fig. 11b). After the decontamination treatment, the sample also had an oxide layer of 300 nm (Fig. 11b) which was believed to be the result of an incomplete ablation of the previous oxide layer or of additional oxidation during the laser cleaning.
In future, cleaning tests will be carried out to compare our results with the literature and to find optimal laser parameters to more efficient decontamination, such as energy per pulse, pulse duration and wavelength or spatial overlap.
 |
Fig. 10 Overview of oxidized 304L SS samples with an initial layer thickness of 3 μm before and after laser cleaning (5.9 J/cm2 · per pulse and 80% of overlapping). |
 |
Fig. 11 GD-MS profiles of a 3 μm thick Eu-contaminated oxide layer on 304L SS (a) before decontamination; (b) after decontamination (18 W, overlapping of 80%, F = 7.6 J/cm2 · per pulse, 1 pass). |
4 Conclusion
Our goal was to develop an oxidation process in order to prepare samples with characteristics similar to those of metal samples oxidized under real nuclear conditions. The creation of a contaminated oxide layer with controlled characteristics is a first step for the study of laser cleaning of metallic surface with micrometric cracks.
The oxidized sample of 304L SS was used to carry out studies with non-radioactive contaminants (Eu) and conventional equipment. These oxides layers were prepared with a contamination by Eu solution spraying, pre-oxidation by laser and a final furnace treatment to continue the oxidation. The thicknesses of the obtained oxides ranged from 0.2 μm up to 4.5 μm with a mean Eu-concentration of 6 × 1016 atoms/cm2 (15 μg/cm2) in the bulk of oxide layer. The method using a furnace treatment simulated the oxidation of stainless steel when it is exposed to high temperature. The resulting oxide layer thickness could be controlled by the set of main parameters (delivered laser energy, duration of the oxidation by furnace or Eu concentration).
It has been shown that laser heating with the parameters of the pre-oxidation led to the formation of a liquid layer of metal in which the Eu contamination could penetrate. The melting of a sub micrometric layer was then needed to fix the contamination the bulk of the layer.
The laser cleaning of the prepared samples resulted in a satisfactory decontamination rate and low residual contamination (≤0.02 wt.%). Future cleaning tests will be carried out to determine laser parameters for more efficient decontamination, such as energy per pulse, pulse duration and wavelength or spatial overlap.
References
- F. Brygo, Ch. Dutouquet, D. Le Guern, R. Oltra, A. Semerok, J.M. Weurlesse, Appl. Surf. Sci. 252, 2131 (2006) [CrossRef] [Google Scholar]
- F. Champonnois, F. Beaumont, C. Lascoutouna, US Patent 20110315666 A1, 2009 [Google Scholar]
- A. Leontyev, Ph.D. thesis, Université Paris Sud − Paris XI, 2011 [Google Scholar]
- Ph. Delaporte, M. Gastaud, W. Marine, M. Sentis, O. Uteza, P. Thouvenot, J.L. Alcatraz, J.M. Le Samedy, D. Blin, Appl. Surf. Sci. 197–198, 826 (2002) [CrossRef] [Google Scholar]
- A. Leontyev, A. Semerok, D. Farcage, P.Y. Thro, C. Grisolia, A. Widdowson, P. Coad, M. Rubel, Fusion Eng. Des. 86, 1728 (2011) [CrossRef] [Google Scholar]
- Z. Homonnay, E. Kuzmann, K. Varga, Z. Nemeth, A. Szabo, K. Rado, K.E. Mako, L. Köver, I. Cserny, D. Varga, J. Toth, J. Schunk, P. Tilky, G. Patek, J. Nucl. Mater. 348, 191 (2006) [CrossRef] [Google Scholar]
- F. Riffard, H. Buscail, E. Caudron, R. Cueff, C. Issartel, S. Perrier, Appl. Surf. Sci. 252, 3697 (2006) [CrossRef] [Google Scholar]
- N. Karimi, Ph.D. thesis, Université Blaise Pascal-Clermont-Ferrand II, 2007 [Google Scholar]
- N. Karimi, H. Buscail, F. Riffard, F. Rabasten R. Cueff, C. Issartel, E. Caudron, S. Perrier, Mater. Sci. Forum 595, 733 (2008) [CrossRef] [Google Scholar]
- M. Wautelet, Appl. Phys. A 50, 131 (1990) [CrossRef] [Google Scholar]
- D. Bäuerle, Chemical Processing with Lasers (Springer, Berlin, 2000) [CrossRef] [Google Scholar]
- P. Lacombe, G. Béranger, B. Baroux, Les aciers inoxydables (Les éditions de physique, 1990) (in French) [Google Scholar]
- A. Semerok, S.V. Fomichev, J.-M. Weulersse, F. Brygo, P.-Y. Thro, C. Grisolia, J. Nucl. Mater. 420, 198 (2012) [CrossRef] [Google Scholar]
- V.P. Veiko, T. Y. Mutin, V.N. Smirnov, E.A. Shakhno, Laser Phys. 21, 608 (2011) [CrossRef] [Google Scholar]
Cite this article as: Luisa Carvalho, Wilfried Pacquentin, Michel Tabarant, Hicham Maskrot, Alexandre Semerok, Growth of micrometric oxide layers to explore laser decontamination of metallic surfaces, EPJ Nuclear Sci. Technol. 3, 30 (2017)
All Tables
Oxide thickness after laser treatment (237 J/cm2 total delivered) and a furnace oxidation (100 h, 873 K) with different contamination concentration in Eu.
All Figures
 |
Fig. 1 Pattern of the scanning regime. The circles represent the consecutive laser spots on the treated surface and the continuous arrow shows the laser beam path. |
| In the text | |
 |
Fig. 2 GD-OES profile of an Eu-contaminated oxide layer on 304L SS after a preliminary laser treatment (9 W, overlapping of 95%, E = 600 J/cm2) and without furnace treatment. The oxide thickness was estimated to be 500 nm. |
| In the text | |
 |
Fig. 3 GD-OES profile of an Eu-contaminated oxide layer on 304L SS after preliminary laser treatment (9 W, overlapping of 95%, E = 600 J/cm2) and oxidation in the furnace (50 h, 873 K). The oxide thickness was estimated to be 4.5 μm. The scale of Eu-distribution was multiplied by 10 in order to be readable. |
| In the text | |
 |
Fig. 4 SEM image (5 000x) of a cross section of a 4.5 μm thick Eu-contaminated oxide layer on 304L SS after a preliminary laser treatment (9 W, overlapping of 95%, E = 600 J/cm2) and oxidation in the furnace (50 h, 873 K). |
| In the text | |
 |
Fig. 5 EDX map of a cross section of a 4.5 μm thick Eu-contaminated oxide on 304L SS after a preliminary laser treatment (9 W, overlapping of 95%, E = 600 J/cm2) and oxidation in the furnace (50 h, 873 K). |
| In the text | |
 |
Fig. 6 Oxide thicknesses resulting from different laser irradiations at 5, 9, 14 and 18 W and 80% overlap (in blue) or 90% overlap (in orange). |
| In the text | |
 |
Fig. 7 SEM image (x 700) of a single impact on 304L SS after a different laser treatment: (a) 9 W (4.1 J/cm2), (b) 14 W (6.3 J/cm2). The red circles represent the thermally affected zone of the sample surface. |
| In the text | |
 |
Fig. 8 Temperature evolution at the surface on the laser beam axis during a single laser pulse of 110 ns for a fluence of 1 J/cm2 on a blank AISI 304L substrate. |
| In the text | |
 |
Fig. 9 Oxide thicknesses for different oxidation durations of Eu-contaminated samples (blue) and blank samples (orange). |
| In the text | |
 |
Fig. 10 Overview of oxidized 304L SS samples with an initial layer thickness of 3 μm before and after laser cleaning (5.9 J/cm2 · per pulse and 80% of overlapping). |
| In the text | |
 |
Fig. 11 GD-MS profiles of a 3 μm thick Eu-contaminated oxide layer on 304L SS (a) before decontamination; (b) after decontamination (18 W, overlapping of 80%, F = 7.6 J/cm2 · per pulse, 1 pass). |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


